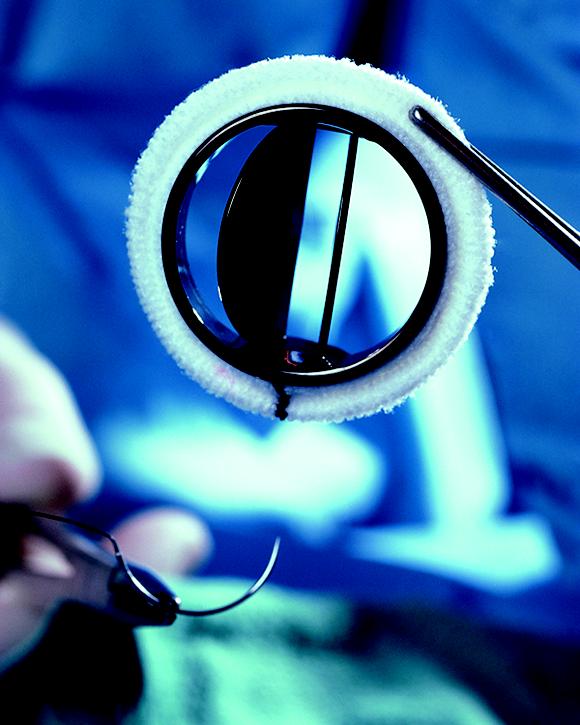
AXF-5Q Graphite
To discuss your specific needs, contact your local Entegris representative.
Don't have an Entegris representative?
Contact UsThank you for requesting a quote. Someone from Entegris will contact you shortly.
Your quote number is {0}.
- Mechanical heart valves
- Orthopedic prosthetics
- Cancer treatments
Specifications
| Particle size | 5 µm (200 µin) |
| Pore size | 0.8 µm (32 µin) |
| Total porosity | 20% volume |
| Open porosity | 80% of total |
| Apparent density | 1.78 g/cm3 (0.064 lb/in3) |
| Compressive strength | 138 MPa (20,000 psi) |
| Flexural strength1 | 86 MPa (12,500 psi) |
| Tensile strength2 | 62 MPa (9,000 psi) |
| Shore hardness | 74 |
| Electrical resistivity | 1470 µΩ-cm (580 µΩ-in) |
| Thermal conductivity | 95 W/m-K (55 Btu-ft/hr/ft2 °F) |
| Oxidation threshold3 | 450°C (840°F) |
| 1Measured using 4-point bend method. | |
| 2Estimated at 70% of flexural strength. | |
| 3Temperature that results in 1% wt. loss in 24 hrs. Oxidation threshold increases by approximately 100°C if graphite is purified. Test sample size equals 0.5” x 0.5” x 1.0". | |
Successfully Opted for software Updates
| Part Number | Material Type |
|---|---|
| DE-18338 | Sample |